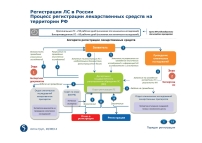
Registration dossier and market authorization
The smooth and prompt registration directly impacts the product launch, as well as achieving the sales and profit targets and the overall brand performance.
Aston Group provides comprehensive support for a new product launch and market entry:
- Preparing registration dossier
- Preparing application for new therapeutic indications
- Building up pharmacoeconomic data to secure reimbursement
- Putting together evidence to include the product in the negative drug lists
- Filing certificate to confirm production pursuant to GMP
Preparing documents to approve GMP manufacturing conditions based on the inspection of the production site:
- Application for GMP Compliance Certificate
- Product evaluation for conformance with quality requirements
- List of products manufactured on the site being inspected
- A letter of consent for inspection from manufacturer
- GMP site file
CJSC Aston Consulting, 1999-2021